Home
A comprehensive resource for safe and responsible laser use
UK: Reports of UFO over Coventry turns out to be light show laser

The blue-green shaft of light was from a laser said to be "the world's most powerful display laser." According to Mat Lawrence from the laser company Kvant, advance notice had been made to the U.K. Civil Aviation Authority before the test was done.
Lawrence said the beam could be seen from 20 miles away.

From The Star and Coventry Live
Australia: Airshow biplane shoots fireworks and laser beams
The lasers start at about 0:27 and last until about 0:55 in the video, which is 3 minutes 12 seconds long. A few times, the lasers appeared to go directly into the crowd — or at least towards the camera that captured the show.

As the biplane lands, starting at about 2:29 the laser projector can be seen as a square black box mounted above the top wing.

The show was created by Scandinavian Airshow. The company says "The Catwalk is the ultimate air show aircraft and can in the evening transform into a pyrotechnic platform and perform its amazing night pyrotechnic display." There is no mention of lasers on their Catwalk webpage as of March 12 2019, so this may be a relatively new addition.
As one YouTube commenter wrote, "That's hella ironic if a plane is shooting lasers at you 😂"
From YouTube, via BGR
India: Laser show attacked as "insult to Lord Shiva" (not a laser)
In an April 28 2018 news story, a Congress party leader said the April 29 - May 4 show on Kedarnath Temple is an insult to Lord Shiva and his devotees.
The 25-minute show is privately funded by the Akshar Travels group of companies, in part to attract visitors and pilgrims to the temple, which is so old its builders and date of construction are not known.
From a YouTube video of the show, it appears there are no lasers used. Instead, the show consists of video projected onto the temple plus narrow lights similar to spotlights or the Clay Paky “Sharpy” moving beam light. The video projector’s light source could possibly be from lasers but even then it is not a “laser show” by the conventional definition of a show using laser beams and/or cartoon-like simple outline graphics.

From The Statesman and the Pioneer
US: FDA recommends against using Laserworld and Ray Technologies laser projectors
Laserworld set up a special website, www.cdrh.info, with a statement and information from their viewpoint.
The International Laser Display Association published guidance for ILDA Members and others who are doing shows in the U.S. with Laserworld and RTI projectors.
US: FDA recalls certain X-Laser light show projectors
FDA listed the following actions:
X-Laser LLC will bring into compliance:
1. All purchasers and associated dealers of affected LLS projector models will be notified by mail and email of their failure to comply with the performance standard. The notification will follow the format and include the information required by 21 CFR 1003.21. Those that do not respond within 14 days will be notified a second time. Those not responding to the second attempt will be notified again every 6 months for the next 2 years. Non-responsive dealers will be ineligible for future orders.
2. All affected LLS projectors will be repaired by removing the auto and music modes from the dipswitch accessible settings, after which, these modes will only be accessible through the DMX connection. These actions, including transportation of the LLS projector, will be made free of charge.
3. All LLS projector models that X-Laser receives, regardless of purpose, will be checked for dipswitch accessible auto or music modes and repaired if needed.
4.Corrective actions will be completed within 120 days of receipt of this letter.
For further questions please call (866) 702-7768.
For additional details from X-Laser, click the “read more” link. Click to read more...
Worldwide: 3D printing creates mechanical "laser show" using a pointer
It works by putting a standard pen-type laser pointer between two cams. Cranking a handle turns the cams which bounce the laser pointer up/down and left/right to create projected patterns:

By using different cam shapes, different patterns can be projected:
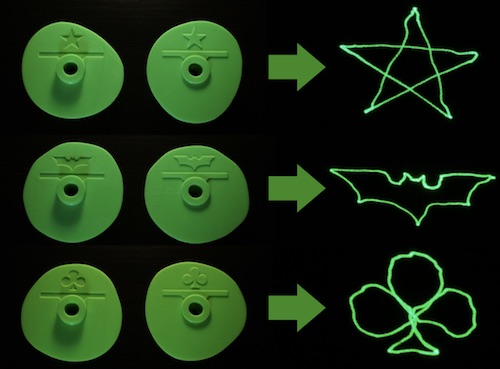
Instructions and plans are available online, including Thingiverse 3D printing files.
Stanford noted “At this point I think it is unlikely I will continue the project. But if I did, here’s what I could do:” He then listed adding blinds to make discontinuous patterns, making the device motor driven, and adding a web service to make it easier to create new cam patterns.
From Evan Stanford’s Hackaday.io page, posted in mid-June 2017
US: Laser show company has variance revoked for unauthorized audience scanning
On July 24 2014, the Food and Drug Administration sent a letter to David Fleenor of Epic FX, Inc. of Phoenix, Arizona. It stated that videos posted on the epicfx.com website “documents audience scanning with Class IIIb and/or Class IV lasers. Although much of the audience scanning was done with fanned beams, your projector is not designed nor reported for safe audience scanning. Your variance prohibits audience scanning. Any laser beams projected into the audience directly or indirectly is considered audience scanning. This is in violation of Condition 5 of your variance.” [The page has since been removed, and returns a 404 error.]
Click to read more...